Specialists dealing with immunization for the Covid-19 assert that testing will be a lot quicker than for a run of the typical vaccine. While some are hopeful that it may be accessible by the end of the year or mid 2021. The speed at which these immunizations are being created is phenomenal, nonetheless effectiveness and safety have become a vital concern to the delivery.
Scientists are trying 48 vaccines in clinical trials on people, and at any rate 88 vaccines are under dynamic persistent examination in preclinical stage, I.e in animals. In China, where the outbreak erupted first, researchers developed five vaccines already on humans, and a government body assures it could be available by the end of this year.
How long does it take to develop a vaccine in general?
Not many known that a vaccine is categorised as a new drug which comes under the protocol of new drug development. There are certain trail phases before the vaccines comes to the market.
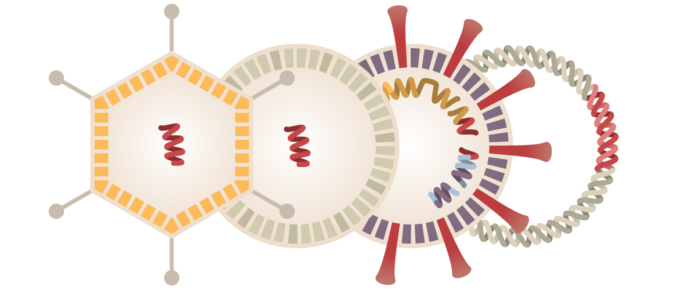
- The initial phase begins with discovery research where a vaccine usually takes approximately 2-5 years to initiate testing on 100 potential vaccines, costing up to $500 million.
- In Phase-0, clinical trials are performed in less than 15 individuals. Investigators use small dosage to make sure that it doesn’t turn harmful before using larger doses in phase 1. If effective results are not seen, investigators proceed to preclinical stage before deciding to continue.
- Next in preclinical stage 20 potential vaccines are tested for 2 years on animals like rodents, Guinea pigs as they mimic the biological response as that of humans.
- This is followed by the phase-1 clinical trials. In this stage, 20-80 healthy human volunteers are enrolled as subjects and tested by the principal investigators (researchers of clinical department) to see for adverse reactions and safety profile.
- Researchers proceed for phase -2 trails, where they check for good immune response from the subjects after injecting an inoculation dose of 5 potential vaccines with new combinations. It uses 100 or more healthy volunteers.
- In phase-3, the investigators usually spend another 2-4 years to narrow down to one potential vaccine to check whether it controls the disease. Third stage will reveal relatively all possible adverse reactions generated from human body in 1000’s of volunteers, which would determine safety and efficacy.
- Subsequently the safety profile, good immune response reports and the potential of the newly developed inoculation data is filed as per the protocol set by respective regulatory body and institutional ethics committee. After submitting, it can take another year for review and approval.
- Manufacturing and delivery requires specialist high end and expensive facilities to produce drugs in bulk. It begins in phase three where 1000 of doses are required to produce.
- After which fourth and final stage, post marketing surveillance commences, where the new vaccine is released in the market. However, the safety profile and any untowardly reactions generated in human are monitored by regulatory body consistently.
COMBINED PHASES: In emergency situations, researchers adopt fast track approvals using combined phases. It is an alternative way to boost up vaccine development. Few coronavirus vaccines are now in Phase 1/2 trials, for instance, in which they are tested for the first time on hundreds of people. This allows the new drug research to compress the timelines to 3-4 years. However scientists claim, it can be done in 1 year too, which is still not proven to be successful yet.
Latest developments across the world
Currently, 34 vaccines are in first phase, 16 in second phase, while 11 are in third phase and 6 in final phase for limited use.
Entos pharma, Israeli institute for Biological research, Immunity Bio, Shenzhen Kangtai are in phase-1. Inovio, DNA based vaccine developed by Angus, India’s Zydus Cadila, Thailand’s Chula Vaccine Research Center, Sanofi etc are in phase 2.

Other genetic vaccines in active preclinical stage include vaccines from: Applied DNA Sciences, EvviVax and Takis Biotech; DIOSynVax; Elixirgen Therapeutics; ETheRNA; HDT Bio; Infectious Disease Research Institute and Amyris; Mediphage Bioceuticals; the OPENCORONA Consortia;
Moderna manufactures vaccine based on messenger RNA (mRNA) to generate viral proteins in the body, which is yet to be launched. In collaboration with National Institutes of Health, in March, the company commenced the first Covid-19 vaccine into human trials, which shown great results. The vaccine is in Phase 3 testing currently.
Currently reports say that Oxford’s Covid-19 vaccine in collaboration with AstraZeneca Plc may get emergency approval before Christmas. Although the final trials are underway, It is likely to get emergency approval, where patients would receive a single vaccine shot.
When can we expect the covid vaccine in India?
In July, the Drug Controller General of India (DCGI) had approved Bharat Biotech to conduct covid-19’s phase 1 and 2 clinical trials.
Bharat Biotech(BB) is keen to release its covid-19 vaccine–Covaxin–by June next year. However, it depends on the government to authorise it for emergency use, based on data from earlier phases of clinical trials, said executive director Sai Prasad.
BB had applied to the DCGI on October 2 seeking approval to conduct phase 3 randomised double-blind placebo-controlled multicentre trial of its COVID-19 vaccine.
Five days ago, the Drugs Controller General of India (DCGI) gave a green signal to Bharat Biotech to conduct phase 3 clinical trials of Covaxin with certain limitations. Reports claim, Covaxin will start its Phase III clinical trials at a private hospital in Bhubaneswar soon.
Similar to previous trials, half of the volunteers would receive placebo and the other half, COVAXIN in this phase. Health care workers would be enrolled for the trial and would be followed up over a certain period of time to asses the efficaciousness of the vaccine in controlling the development of the virus disease, Dr Venkata Rao, Professor in the department of Community Medicine at the Institute of Medical Sciences and SUM Hospital said.
Cadila Healthcare Ltd., an Ahmedabad based firm, is one of two Indian drugmakers accelerating to develop an indigenous Covid-19 vaccine. It is in discussions with potential partners to boost the production, if its candidate gets through human clinical trials.
The makers are eyeing to rope in contract manufacturers for an additional 50 million to 70 million doses of its plasmid DNA vaccine according to Managing Director Sharvil Patel.
Cadila, which also delivers coronavirus treatments through remdesivir, is among a number of Indian companies conducting vaccine trials. It is currently in second stage and If the results are promising, Cadila will start recruitment of more than 30000 subjects in December final-stage clinical trials. Patel said, the process could take two to three months and that he hopes to see faster approvals from Indian regulators.
After a preliminary data of vaccine stages, it is not still clear how the vaccine makers are assuring the public of its vaccine by year end or mid 2021. It is to be noted that several vaccines which developed ranging from 5-6 years too failed previously to deliver consistent successful results. In this present scenario, these two firms just began trials from March or April 2020 in India.
Comparatively Bharat Biotech could possible launch the vaccine by January or February and Zadilla by March or April in 2021. Although the vaccine is released in the market, there is no guarantee that it would show good and effective results. Experts claim that such inoculation doses needed to be distributed in two to three shots for a considerable time in large population and then only it can be assessed if the vaccine is a success or failure.